The most efficient use of space however is obtained in the two close-packed structures, the face-centred cubic (fcc) and the hexagonal close-packed (hcp) structures, which are both based on hexagonal layers and take up 74% of the space.
The face-centred cubic and hexagonal close-packed structures have some similarities:
- Both are based upon stacking layers of atoms, where the atoms are arranged in a close-packed hexagonal manner within the individual layer.
- The atoms of the next layer of the structure will sit in some of the hollows in the first layer – this provides the closest approach of atoms in the two layers and hence, maximizes the cohesive interaction. The position of the next layer of atoms determines if the structure is face-centred or hexagonal close-packed.
- Each atom has 12 nearest neighbours. = coordination number is 12.
Face-Centred Cubic
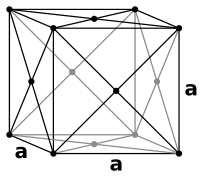
The atoms of the 3rd layer are laterally offset from those in both the 1st and 2nd layers. It is not until the 4th layer that the sequence begins to repeat. This is the ABCABC packing sequence of the fcc structure.
The face-centred cubic (fcc) structure is the same as the ordinary simple cubic lattice with the addition of an extra atom at the centre of each cube face. The close-packed layers are the body diagonal planes of the cube.
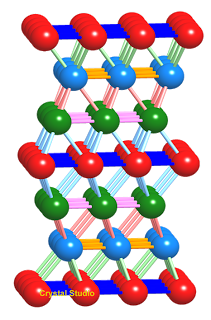
Ionic bonds, unlike covalent bonds, are not directional and do not saturate, and so the ions can pack together as closely as possible. A sodium chloride crystal consists of Cl– ions arranged in the face-centred cubic lattice, with the smaller Na+ ions fitting in the gaps.