Table of Contents
Electromagnetic Waves
In terms of classical wave theory, a very hot object such as any star or sun produces a range of electromagnetic waves. (E.g. The Sun produces ultraviolet light in addition to visible light) Electromagnetic waves are often collectively referred to as electromagnetic radiation.
Electromagnetic radiation is produced by acceleration or sudden movement of electrons. The electron in motion constitutes the electric current that generates the magnetic field in the wave.
- Electromagnetic waves are transverse waves, consisting of electric and magnetic waves at right angles to each other and perpendicular to the direction of wave propagation.
- Electromagnetic waves can travel through vacuum and do not need a medium to traverse. They can travel through solids, liquids and gases.
- All electromagnetic waves have the same speed in vacuum of 3 x 108 m s-1 (commonly referred to as the ‘speed of light’). But the speed slows down in other material media.
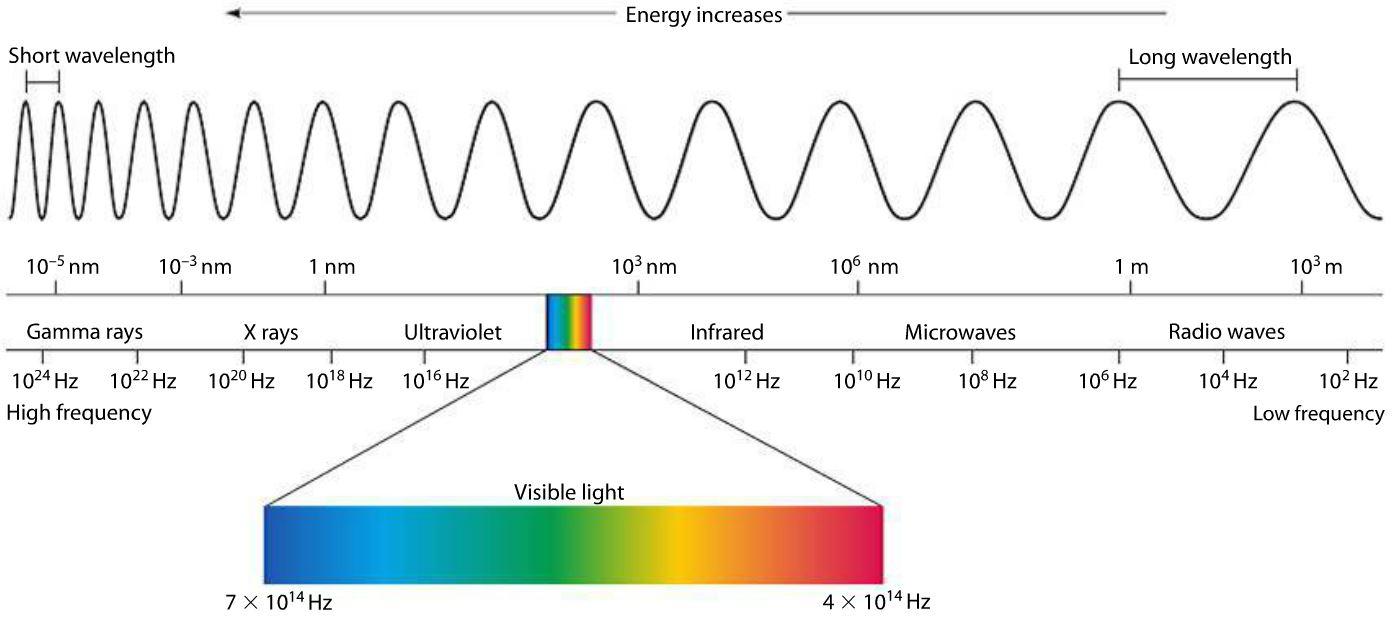
Electromagnetic Spectrum
The electromagnetic spectrum encompasses a vast range of radiation types, each with unique properties and applications, distinguished by their frequency and wavelength. This spectrum is continuous due to the myriad of oscillators present in any material, which can emit radiation across all frequencies.
Gamma Rays
Gamma rays are the most energetic forms of electromagnetic radiation, with frequencies above $10^{19}$ Hz and wavelengths less than $10^{-11}$ meters. They possess the ability to penetrate most materials easily, making them useful in medical imaging and treatments, as well as in industrial applications for material inspection. Gamma rays are typically produced by nuclear reactions, such as those occurring in radioactive decay or in nuclear explosions.
Hint: To convert between frequencies and wavelengths, employ the wave velocity formula $v = f \lambda$, where $v$ represents the wave’s speed. In the context of gamma rays traveling through a vacuum, this speed aligns with the speed of light, approximately $3 \times 10^8$ meters per second, offering a precise benchmark for conversions.
Applications: Applications Of Gamma Rays
X-Rays
Directly following gamma rays in energy, X-rays have frequencies ranging from $10^{17}$ to $10^{19}$ Hz and wavelengths between $10^{-11}$ to $10^{-8}$ meters. They are known for their ability to pass through soft tissue but are absorbed by denser materials like bone, which is why they are extensively used in medical imaging. X-rays are produced by X-ray tubes, which involve accelerating electrons to high energies before colliding them with a metal target.
Applications: Applications Of X-Rays
Ultraviolet (UV) Radiation
Ultraviolet radiation spans frequencies from about $10^{15}$ to $10^{17}$ Hz, with wavelengths from $10^{-8} to $10^{-7}$ meters. Produced by very hot bodies, such as the Sun, UV radiation is mostly filtered by Earth’s atmosphere but is responsible for phenomena like sunburn. It plays a crucial role in vitamin D synthesis in humans but can also cause damage to living tissue and materials over time.
Applications: Applications Of Ultraviolet
Visible Light
Visible light, the only part of the spectrum we can see, ranges from 430-770 terahertz (THz) in frequency and 390 to 700 nanometers (nm) in wavelength. It encompasses the colors violet, indigo, blue, green, yellow, orange, and red, in order of decreasing frequency. The Sun and other similar stars emit a broad spectrum of light that appears white to the human eye, but can be dispersed into its constituent colors by a prism.
Applications: Applications Of Visible Light
Infra-Red (IR) Waves
Infrared radiation follows visible light in the spectrum, with frequencies from about 300 GHz to 430 THz and wavelengths from 700 nm to 1 mm. It is emitted by all objects based on their temperatures, with hotter bodies emitting more IR. This radiation is widely used in heaters, cooking, and infrared cameras for night vision and thermal imaging.
Applications: Applications Of Infra-Red Waves
Microwaves
Microwaves occupy frequencies between 1 GHz to 300 GHz, with wavelengths in the range of 1 mm to 30 cm. They are generated by devices like klystron valves and magnetrons and are used in radar, telecommunications, and notably for cooking in microwave ovens. Microwaves interact with materials by causing water molecules to vibrate, producing heat.
Applications: Applications Of Microwaves
Radio Waves
With the longest wavelengths in the electromagnetic spectrum, radio waves range from a few centimeters to thousands of meters and have frequencies from 30 Hz to 300 GHz. They are produced by oscillations in electronic circuits and are used in all forms of wireless communications, including radio, television, and cell phones. Radio waves are easily generated and can travel long distances, making them ideal for communication.
Each portion of the electromagnetic spectrum has unique properties and uses, from the high-energy gamma rays and X-rays used in medical and industrial applications, through visible light that enables our vision, to radio waves that form the backbone of modern communication systems. Understanding the spectrum’s diversity is crucial for harnessing its potential across various scientific and technological fields.
Applications: Applications Of Radio Waves
Overview of Electromagnetic Spectrum Properties and Applications
| Type | Frequency Range | Wavelength Range | Production | Common Uses | Properties |
|---|---|---|---|---|---|
| Gamma Rays | > 1019 Hz | < 10-11 meters | Nuclear reactions | Medical imaging, industrial inspection | High penetration, very high energy |
| X-Rays | 1017 to 1019 Hz | 10-11 to 10-8 meters | X-ray tubes | Medical imaging, security | Penetrates soft tissue, absorbed by bone |
| Ultraviolet (UV) | 1015 to 1017 Hz | 10-8 to 10-7 meters | Hot bodies like the Sun | Vitamin D synthesis, sterilization | Filtered by atmosphere, causes sunburn |
| Visible Light | 430 to 770 THz | 390 to 700 nm | Sun, various light sources | Seeing, photography, illumination | Only spectrum visible to humans |
| Infrared (IR) | 300 GHz to 430 THz | 700 nm to 1 mm | Hot objects | Heating, night vision, remote controls | Emitted by all objects, heats objects |
| Microwaves | 1 GHz to 300 GHz | 1 mm to 30 cm | Klystron valves, magnetrons | Communication, radar, cooking | Heats water molecules, reflected by metals |
| Radio Waves | 30 Hz to 300 GHz | 1 cm to thousands of meters | Electronic circuits | Communication (radio, TV, cell phones) | Long distance travel, easily generated |
Worked Examples
Example 1: Understanding Wave Properties
If a scientist observes an electromagnetic wave with a frequency of $5 \times 10^{14}$ Hz, in which part of the electromagnetic spectrum does this frequency fall, and what are some possible sources or uses of this type of radiation?
Click here to show/hide answer
The frequency of $5 \times 10^{14}$ Hz falls within the visible light range of the electromagnetic spectrum. This type of radiation could be emitted by the sun or other white-hot bodies as visible light. It is the only part of the electromagnetic spectrum that is visible to the human eye and is used in a wide range of applications including illumination, photography, and as a signal in optical communication technologies.
Example 2: Wave Behavior in Different Mediums
Explain why electromagnetic waves can travel through the vacuum of space, but sound waves cannot.
Click here to show/hide answer
Electromagnetic waves, such as light and radio waves, can travel through the vacuum of space because they do not require a medium to propagate. They consist of oscillating electric and magnetic fields that support each other as they travel through space. On the other hand, sound waves are mechanical waves that require a medium (solid, liquid, or gas) to travel through, as they propagate through the vibrations of particles in the medium. In the vacuum of space, where there are no particles to vibrate, sound waves cannot travel.
Example 3: Safety and Electromagnetic Radiation
Considering the electromagnetic spectrum, why is it safe to be exposed to radio waves and infrared radiation but not to UV radiation or X-rays for extended periods?
Click here to show/hide answer
Safety regarding exposure to different types of electromagnetic radiation depends on the energy of the waves. Radio waves and infrared radiation have much longer wavelengths and, consequently, lower energy levels than UV radiation or X-rays. Lower energy electromagnetic waves like radio and infrared are generally safe and do not have enough energy to ionize atoms or disrupt molecular structures significantly in living tissues. On the other hand, UV radiation and X-rays have much shorter wavelengths and higher energy levels, capable of ionizing atoms and causing damage to living cells and DNA, leading to burns, cancer, and other health issues. Therefore, prolonged exposure to high-energy electromagnetic radiation like UV rays and X-rays is hazardous to health.