Classical View
- Radiation is wavelike in nature
- When radiation is incident on an object, the particles near the surface will absorb the radiation and becomes “excited”
- The excited particles will emit radiation to lose the gained energy and regain stability
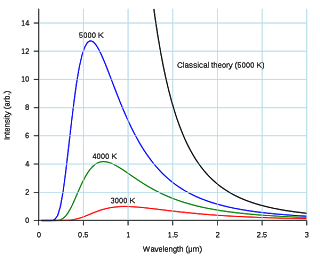
Using the above classical view, physicists attempted to explain blackbody radiation. One of the calculations yield Rayleigh-Jeans Law: $I \left( \lambda, T \right) = \frac{2 \pi c k_{b} T}{\lambda^{4}}$
- At long wavelengths, the calculations are in good agreements with the experimental data
- However, according to the equation, as wavelength approaches zero, the intensity of the radiation approaches infinity. = Energy emitted by a blackbody becomes infinite as the wavelength limit approaches zero. (FAIL) (Known as ultraviolet catastrophe)
- Experimental data: Wavelength approaches zero, emitted radiation intensity approaches zero
This figure shows the failure of Rayleigh-Jeans Law in predicting the intensity of emitted radiation at wavelengths approaching 0.