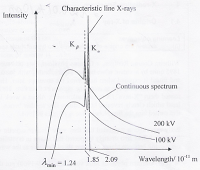
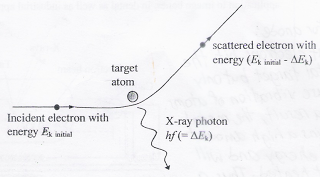
The continuous spectrum and the peaks of X-rays are produced from two processes:
- When fast-moving electrons emitted from the cathode are suddenly decelerated inside the target anode – these rays are called bremsstrahlung radiation, or “braking radiation”.
- When electrons in the target atoms get ‘excited’ and then ‘de-excited’ – X-rays produced in this way have definite energies just like other line spectra from atomic electrons. They are called characteristic X-rays since their energies are determined by the atomic energy levels which they transit.
Explanation For Continuous X-ray Spectrum:
An incident electron with energy Ek initial collides with a target atom. The fast moving electron interacts with the nucleus of the target atom and as the electron approaches the nucleus, it is slowed down. The loss of kinetic energy is converted into X-ray photons that are emitted. The energy of the photon released depends on how close the negative electron comes into contact with the positive nucleus. The closer an electron approaches the nucleus, the higher the energy of the released photon.
The scattered electron which now has energy less than Ek initial may have a subsequent collision with another target atom, generating a second X-ray photon. The electron-scattering process continues until the electrons is approximately stationary, loses all its energy. All the X-ray photons generated from these collisions between electrons and target atoms form part of the continuous X-ray spectrum.
The minimum wavelength can be explained by a collision in which an incident electron stops abruptly because the kinetic energy of the electron is completely converted into an X-ray photon (with maximum photon energy).
Total kinetic energy of fast moving electron = Maximum Energy of a X-ray photon
eV = Energy of X-ray photon
$ \frac{1}{2}m_{e}V_{e}^{2} = \frac{hc}{\lambda_{min}}$
Explanation for Characteristic X-ray Spectrum
An accelerated electron from the cathode collides into an electron of a target atom that is orbiting in the K-shell(n=1). If sufficient energy is transferred by the accelerated electron to the orbiting electron, the latter electron will be ejected from the target atom.
Steps for emission of Kɑ
- Electron is ejected from K-shell (n=1) after collision by an accelerated electron from the cathode
- Electron from L-shell (n=2) drops down to K-shell (n=1)
- An X-ray photon of Kɑ characteristic X-ray is emitted
Steps for emission of Kβ
- Electron is ejected from K-shell (n=1) after collision by an accelerated electron from the cathode
- Electron from M-shell (n=3) drops down to K-shell (n=1)
- An X-ray photon of Kβ characteristic X-ray is emitted
The rates of emission of the Kɑ and Kβ characteristic X-rays are high, hence their intensities are high. The intensity of the Kɑ characteristic X-ray is typically greater than the Kβ characteristic X-ray. This is because the electrons in the L-shell are closer to the K-shell, hence there is a greater probability that the vacancy in the K-shell is filled by an electron from the L-shell than from the M-shell.
Since the energy differences between electrons in the various energy levels is a characteristic of the target atom, the wavelength of the Kɑ and Kβ characteristic X-rays are unique for each element. The wavelength of the characteristic X-ray is shorter for elements of higher proton number.
When the accelerating potential V is increased from 100kV to 200kV, the minimum wavelength of the X-rays, λmin is shorter, but the wavelengths of the characteristic X-rays remain unchanged. The intensity of all wavelengths increases.