Table of Contents
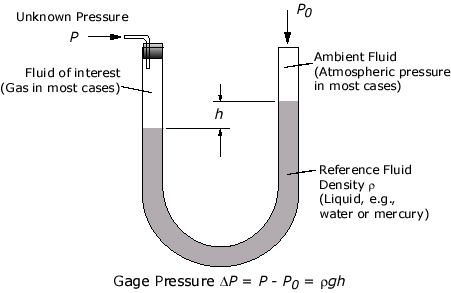
Manometer
A differential manometer is a simple instrument for comparing pressures, usually by the difference in height of two liquid columns.
- The simplest of such instrument is a U-tube containing some liquid, usually mercury, water or oil.
- The pressure exerted by a confined gas changes the levels of the mercury in the manometer. The total pressure of a gas or liquid is the total pressure it exerts, including the effect of atmospheric pressure. ($p_{atm}$).
Changes To Liquid Levels In Manometer
When the manometer is not connected to any gas supply, the liquid levels in the left and right side of the are equal since atmospheric pressure acts on both surfaces of the liquid.
When one end of the tube is connected to a gas supply, the pressure exerted by the gas changes the levels of the mercury column.
Note: If you are unsure of the computation of pressure due to mercury column, please refer to hydrostatic pressure.
If the gas pressure (from the gas supply) is greater than the atmospheric pressure (as shown by the image above), it will drive down the liquid level in the left side of the manometer. The pressure of the gas, $P_{gas}$ can be calculated by:
$$\begin{aligned} P_{gas} &= \text{atm. pressure } + \text{ pressure of mercury column} (\text{height}, \Delta h) \\ P_{gas} &= p_{atm} + \rho g \Delta h \end{aligned}$$
If the gas pressure (from the gas supply) is smaller than the atmospheric pressure, it will drive down the liquid level in the right side of the manometer. The pressure of the gas, $P_{gas}$ can be calculated by:
$$\begin{aligned} P_{gas} &= \text{atm. pressure }-\text{ pressure of mercury column} (\text{height}, \Delta h) \\ P_{gas} &= p_{atm}-\rho g \Delta h \end{aligned}$$
Worked Examples
Example 1
A manometer is connected to a gas supply.
Pressure can be measured in cm of water.
What is the pressure of the gas?
- 8 cm of water more than atmospheric pressure
- 12 cm of water more than atmospheric pressure
- 8 cm of water less than atmospheric pressure
- 12 cm of water less than atmospheric pressure
Click here to show/hide answer
Answer: A