Table of Contents
A thermometer is useless without a temperature scale (the markings on the thermometer). In order to define a temperature scale, the following steps are required:
- Choose a thermometric substance with a suitable thermometric properties.
- Select two fixed points which are easily obtainable and reproducible. (Fixed points are explained in more details below.)
- Divide the temperature range between the two fixed points into equal divisions.
Fixed Points
- Fixed points are used in calibrating thermometers. To calibrate a thermometer is to mark a thermometer so that you can use it to measure temperature accurately. A fixed point is a standard degree of hotness or coldness such as the melting point of ice or boiling point of water.
- This method of using two fixed points to calibrate a thermometer assumes that temperature changes linearly with the thermometric property. This means that the physical property (that is chosen) changes evenly and regularly with temperature and if we draw a graph of temperature against the thermometric property, we should get a straight-line graph.
Centigrade Scale
The two fixed points used in the Centigrade scale are:
- Steam point (upper fixed point) – The temperature at which pure water boils at one atmospheric pressure and is assigned the value of 100 °C.
- Ice point (lower fixed point) – The temperature at which pure ice melts at one atmospheric pressure and is assigned the value of 0 °C.
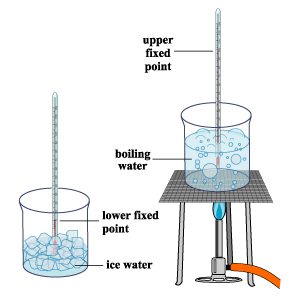
Determination Of Ice Point
- The bulb of the thermometer is immersed into a container filled with pure melting ice. In order to ensure that the contact between the bulb and ice is good, ice shavings can be used.
- When the level indicated by the thermometric substance remains steady after some time, a mark will be made at that point. This mark will corresponds to the ice point (lower fixed point) and is assigned the value of 0 °C.
Determination Of Steam Point
- The bulb of the thermometer should be placed into a container filled with pure boiling water OR placed just above the container (such that it is in contact with the emerging steam). It is important to ensure that the pressure of the environment/room is the same as the atmospheric pressure outside of the environment/room. Different atmospheric conditions will result in the thermometer showing different/wrong temperature readings.
- When the level indicated by the thermometric substance remains steady after some time, a mark will be made at that point. This mark will corresponds to the steam point (upper fixed point) and is assigned the value of 100 °C.
For the Centigrade scale, the distance between the ice point and the steam point is divided into 100 equal parts. Each mark on the thermometer is a measure of 1 °C.
Calculation Of Temperature On Centigrade Scale
- Use an unmarked mercury thermometer
- Place the thermometer just above pure boiling water(upper fixed point) and record its length of mercury thread, l100°, from a reference level (such as base of the reservoir). Repeat the same for the pure melting ice (lower fixed point) and record its length of mercury thread, l0°.
- Now place the same thermometer in an unknown temperature, θ°C and record its length of mercury thread, lθ°.
- The temperature θ°C can be calculated by simple proportion: $$\begin{aligned} \theta^{\circ}\text{C} &= \frac{X_{\theta^{\circ}} – X_{0^{\circ}}}{X_{100^{\circ}} – X_{0^{\circ}}} \: \times \: 100^{\circ}\text{C} \: \\ &= \:\frac{l_{\theta^{\circ}} – l_{0^{\circ}}}{l_{100^{\circ}} – l_{0^{\circ}}} \: \times \: 100^{\circ}\text{C} \end{aligned}$$
- Note: X is the thermometric property which varies uniformly with temperature. In the case of a laboratory thermometer or clinical thermometer, X will be the length of the mercury thread.
Kelvin (Absolute) Scale OR Thermodynamic Scale
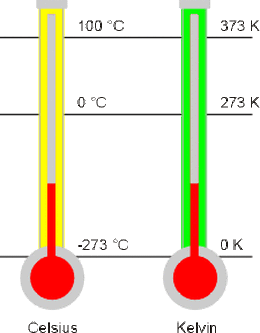
The figure above shows the comparison between the Celcius scale (centigrade scale) and the Kelvin scale.
The unit for the Kelvin Scale is the Kelvin (K), which is the SI unit for temperature.
Kelvin scale or Thermodynamic scale is based on 2 fixed points:
- Absolute zero
- Triple point of water – The triple point of water is the temperature at which saturated water vapour, pure water and ice all coexist in equilibrium (at 0.01 °C and 611.73 Pa)
1 K is defined as the $\frac{1}{273.16}$ of the temperature of the triple point of water.
The intervals on both Kelvin and Celsius scales are the same, such that a difference of 1 K is equivalent to a difference of 1 °C.
You can convert temperature from Celcius scale (centigrade scale) to Kelvin scale and vice versa by using:
$$\theta/^{\circ}\text{C} = T/\text{K}-273.15$$
| Temperature | Kelvin Scale | Celcius Scale |
|---|---|---|
| Absolute Zero | 0 K | $-273^{\circ}\text{C}$ |
| Ice Point | 273 K | $0^{\circ}\text{C}$ |
| Steam Point | 373 K | $100^{\circ}\text{C}$ |
Note that absolute zero is defined as the coldest possible temperature whereby all the particle motion stops.