Reversible Thermodynamic Processes
A reversible transformation is an idealised transformation in which the system is in (very nearly) thermodynamic equilibrium throughout the transformation.
– Therefore the system has well-defined values for all its state variables throughout the transformation.
– The transformation can be plotted and thus followed on a diagram of state variables.
– The system can return to its initial state along the same path by reversing the direction of transformation.
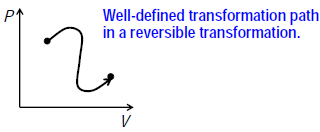 State variables are thermodynamic properties (such as T, P, V and internal energy) of a system. The values of these properties depend only on the state of the system.
State variables are thermodynamic properties (such as T, P, V and internal energy) of a system. The values of these properties depend only on the state of the system.
Equilibrium: When the system is in thermodynamic equilibrium, its properties are unchanging in time.
To achieve a reversible transformation in practice:
- The transformation must happen slowly enough that the system passes through a continuous sequence of equilibrium states. Hence, all reversible processes are quasi-static (i.e. infinitely slow) processes. How slow is enough? This depends on the type of transformation: isothermal processes have to be really very very slow but adiabatic processes can be quite fast, and still behave reversibly.
- There must also be no friction or other dissipation effects. Therefore not all quasi-static processes are reversible.
Irreversible Thermodynamic Processes
A process that is not reversible is irreversible. During an irreversible transformation, the system is in disequilibrium and have no well-defined set of values for its state variables. The transformation cannot be followed throughout on a graph of state variables.
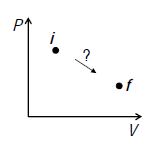 An irreversible process has well-defined initial and final states but no well-defined transformation path.
An irreversible process has well-defined initial and final states but no well-defined transformation path.
Are real processes reversible or irreversible?
Most real processes are irreversible.
– All processes involving heat flow due to a finite temperature difference are irreversible
– All naturally-occurring processes are irreversible
– All processes that convert heat into mechanical work or vice versa are irreversible.
However, a few processes can approach the reversible idealisation pretty well. e.g. adiabatic processes.
So, why do we study the thermodynamics of reversible processes?
They are easy to study! The results set an upper limit to the efficiency of real processes, and they provide insights into how to make real processes more efficient.