Table of Contents
Conduction
Conduction is the transfer of thermal energy from one place to another without any flow of the material medium.
Thermal Conductors & Thermal Insulators
Metals are good thermal conductors.
- Imagine a long rod is heated at one end. Heat travels from the heated end to the cold end via conduction.
- The cold end is heated up faster (i.e. thermal energy is conducted through the rod faster) if the rod has a large cross-sectional area, is short and has a large temperature difference between its ends.
Non-metals are poor thermal conductors.(they are thermal insulators)
- Examples: Wood, glass, plastics, fabrics, cork
Good thermal conductors (e.g. metals) feel colder than thermal insulators (e.g. plastics) in an air-conditioned room, even though they are at the SAME temperature.
- Reason: The metals conduct heat away from the hand faster than the plastics.
- Full article on this: Why Do Metals Feel Colder Than Plastics In Air-con?
Conduction typically requires the objects to be in physical contact with one another.
Mechanisms Of Conduction
1. Atomic Collisions
- In solids, atoms or molecules vibrate about their fixed position. In a hotter region, atoms or molecules vibrate more vigorously or have more kinetic energy than those in the colder region.
- These molecules collide with their neighbours and transfer some of their kinetic energy to them. The neighbours collide with their neighbours. In this way, heat is conducted to colder regions and raised the temperature.
- This is a very slow process.
- Solids conduct heat better than liquids and gases due to their closely packed molecules.
Detailed Explanation
In solids, such as metals or non-metallic crystals, atoms or molecules are arranged in a fixed lattice structure. When heat is applied to a solid, these particles vibrate about their fixed positions. The energy from this vibrational motion is known as kinetic energy. In regions with higher temperatures, the particles vibrate more vigorously, possessing greater kinetic energy compared to those in colder regions.
As the high-energy particles collide with their neighboring particles, they transfer some of their kinetic energy to them. These neighboring particles, in turn, experience an increase in kinetic energy and begin to vibrate more vigorously. This process continues as the energy is passed from one particle to another through a series of collisions, eventually leading to the propagation of heat through the material.
This process is relatively slow, as each individual collision takes time. The effectiveness of heat conduction depends on the nature of the material.
In summary, heat conduction in solids involves the transfer of kinetic energy through the vibrational motion of closely packed particles. This process occurs through a series of collisions that gradually propagate heat from hot to cold regions within the material. The efficiency of heat conduction is influenced by the nature of the material, with solids generally exhibiting better heat-conducting properties compared to liquids and gases.
2. Free Electron Diffusion
- On top of atomic collisions, most metals are known as thermal conductors due to their huge number of free electrons available for thermal conduction. The migration of fast-moving electrons is known as free electron diffusion.
- In solid thermal insulators, the absence of free electrons restricts thermal conduction to the vibrations of atoms and molecules within the crystal lattices.
- This is a very quick process.
Detailed Explanation
In addition to atomic collisions, the exceptional thermal conductivity exhibited by most metals can be attributed to a distinctive feature: the abundance of free electrons available for thermal conduction. In the context of heat transfer, the migration of these fast-moving electrons is referred to as free electron diffusion.
Metals possess a unique electronic structure characterized by a sea of delocalized electrons that are not bound to specific atoms but instead move freely throughout the metal lattice. When heat is applied to a metal, these numerous free electrons contribute significantly to the rapid transmission of thermal energy. As high-energy electrons move through the lattice, they collide with other particles, transferring their kinetic energy and effectively carrying heat across the material.
On the contrary, in solid thermal insulators, the absence of free electrons sets the stage for a different thermal conduction mechanism. In these insulating materials, thermal energy transfer primarily relies on the vibrations of atoms and molecules within the crystal lattices. Without the aid of free electrons, the process of thermal conduction in insulators is comparatively slower.
The crystal lattice structure in insulators restricts the movement of particles to vibrational motion, and the lack of a significant population of free electrons impedes the quick transmission of heat. As a consequence, the thermal conductivity of solid insulators is generally lower compared to metals.
It’s important to note that while free electron diffusion in metals facilitates rapid thermal conduction, the absence of free electrons in insulators confines heat transfer to the relatively slower vibrational motion of atoms and molecules within their ordered lattice structures. This fundamental difference in thermal conductivity mechanisms contributes to the diverse thermal properties observed in different materials.
Applications Of Conduction
Uses Of Good Thermal Conductors
Cooking pans are often made with metals because of their good thermal conduction property. In contrast, the handles of the cooking utensils are made up of insulators to protect the hands from scalding.
Uses Of Poor Thermal Conductors (Thermal Insulators)
Sawdust is used to cover the ice cubes from melting.
A polystyrene cup aids in maintaining the temperature of hot liquids or preserving the coolness of cold liquids. Certain saucepans feature handles crafted from wood or plastic, while cork is employed for table mats. These materials are insulators, exhibiting a slow transfer of thermal energy.
Air stands out as an inefficient thermal conductor, making it an excellent insulator. This property explains why residences equipped with cavity walls (consisting of two layers of bricks separated by an air space) and double-glazed windows maintain warmth during winter and coolness in summer.
Due to air’s poor conductivity, materials that trap air, such as wool, felt, fur, feathers, polystyrene foam, and fiberglass, also exhibit low thermal conductivity. Consequently, these materials serve as effective thermal insulators, applied in insulating water pipes, hot water cylinders, ovens, refrigerators, as well as the walls and roofs of buildings. Additionally, they find use in crafting warm winter apparel, such as fleece jackets.
Divers and water skiers wear wet suits to maintain warmth. The suit becomes wet, creating a layer of water between the individual’s body and the suit. This water, warmed by body heat, retains its warmth due to the insulating properties of the suit, typically crafted from materials like neoprene, a synthetic rubber.
Conduction In Liquids & Gases
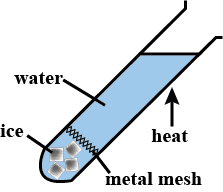
Conduction within liquids and gases occurs at a notably slow pace. Unlike solids, liquids, and gases exhibit a lower capacity for conducting thermal energy.
Water has poor thermal conductivity – i.e. is a poor thermal conductor. The experiment depicted above involves boiling the water at the top of the tube, while the ice at the bottom remains unmelted.
Solids, with their closely packed and well-ordered structure, are generally better conductors of heat compared to liquids and gases.
In liquids and gases, the particles exhibit a lower density and a more disordered arrangement compared to solids. The absence of a regularly ordered lattice structure hampers the establishment of efficient lattice vibrations. Additionally, the lack of free electrons further contributes to their generally inferior thermal conductivity compared to solids. As a result, the transfer of kinetic energy through collisions in liquids and gases is less efficient, leading to poorer heat conduction. In contrast, these states of matter tend to rely more on mechanisms such as convection and radiation for the effective transfer of heat.
Worked Examples
Example 1
Outline an experiment illustrating the characteristics of materials with efficient and inefficient thermal conductivity.
Click here to show/hide answer
Various materials, such as metal, wood, plastic, and glass rods, are cut into uniform lengths and equipped with temperature sensors at one end. The materials are then wrapped in insulating materials to minimize heat loss to the surroundings. A controlled environment is set up, and the exposed ends of the materials are simultaneously heated using a consistent heat source. Temperature measurements at regular intervals allow for the plotting of temperature-time graphs, enabling the comparison of thermal conductivity. Materials exhibiting a rapid temperature increase are identified as good thermal conductors, while those with slower temperature changes are considered poor thermal conductors.
Detailed Description
Objective: The objective of this experiment is to illustrate and compare the thermal conductivity of materials, distinguishing between good and bad thermal conductors.
Materials:
- Various materials with different thermal conductivities (e.g., metal rod, wooden rod, plastic rod, glass rod)
- Insulating materials (e.g., foam or rubber sheets)
- Thermocouples or temperature sensors
- Heat source (e.g., hot plate or Bunsen burner)
- Stopwatch or timer
- Ruler or calipers
- Protective gear (gloves, safety goggles)
Procedure:
- Set up a controlled environment with minimal heat loss, such as a laboratory bench.
- Cut the materials into uniform lengths (e.g., 20 cm) using a ruler or calipers.
- Place a thermocouple or temperature sensor at one end of each material. Ensure that the sensors are securely attached and make good thermal contact with the materials.
- Arrange the materials in a row, leaving spaces in between to prevent heat transfer between them.
- Wrap each material with insulating materials, leaving only the ends exposed where the temperature sensors are located. This ensures that heat is transferred predominantly through the materials and not lost to the surroundings.
- Record the initial temperature of each material using the thermocouples or temperature sensors.
- Heat one end of each material simultaneously using the heat source. Apply the same amount of heat to each material for the same duration.
- Measure and record the temperatures at the exposed ends of the materials at regular intervals (e.g., every 30 seconds) using the thermocouples or temperature sensors.
- Plot a graph of temperature versus time for each material.
- Analyze the results. Materials that experience a rapid increase in temperature are good thermal conductors, while those with slower temperature increases are poor thermal conductors.
- Compare the thermal conductivity of the materials based on the slopes of their temperature-time graphs. The steeper the slope, the better the thermal conductor.
- Draw conclusions about the thermal properties of the materials tested and relate them to real-world applications.