Historical note: Crisis for the equipartition theorem at the end of the 1800s.
The prediction of the equipartition theorem does not agree with the measured heat capacity of gases.
- Boltzmann: Defended the derivation of his equipartition theorem as correct, but suggested that gases might not be in thermal equilibrium.
- Lord Kelvin: Suggested that the derivation for the equipartition theorem must be incorrect, since it disagreed with experiment, but was unable to show how.
- Lord Rayleigh: Suggested that both the equipartition theorem and the experimental assumption of thermal equilibrium were both correct; to reconcile them, he noted that a “new principle” must be found to “escape from the destructive simplicity” of the equipartition theorem.
- Einstein finally provided that escape in 1907 by showing that the anomaly was due to quantum effects: At low temperatures, some modes become “frozen” and no longer available to absorb energy.
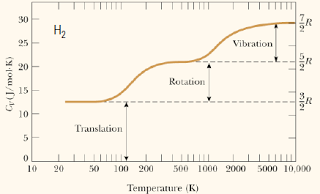
Temperature dependence of Cv
– Rotational motion and then vibrational motion gets activated at increasing temperature.
Hence, at rotational mode, as temperature increase, the molecule do not vibrate harder. There is no energy used to increase the vibrational energy of the molecules. Hence, the vibrational mode do not contribute to the heat capacity at low temperature.