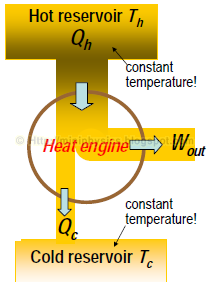
First law for an engine cycle says:
Wout = Qh – Qc
,where
Wout is work output by engine
Qh is heat input from hot reservoir
Qc is heat output to cold reservoir
Thermal efficiency is defined to be the ratio of work output to the heat input from the hot reservoir per cycle,
$$\begin{aligned} e \, &= \frac{W_{\text{out}}}{Q_{h}} \\ &= \frac{Q_{h} \, – Q_{c}}{Q_{h}} \\ &= 1 \, – \frac{Q_{c}}{Q_{h}} \end{aligned}$$
This thermal efficiency is simply 1 minus the fraction of heat rejected into the cold reservoir.
Heat Pump: Coefficients of performance
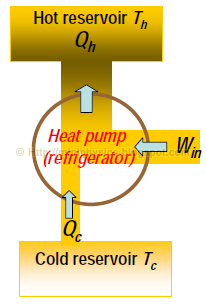
First law for a heat pump is:
$$W_{\text{in}} = Q_{h} – Q_{c}$$
Here, Qh is defined as heat output to hot reservoir and Qc is heat input from cold reservoir, because we are dealing with a heat pump.
In the heating mode, where we are interested in the ratio of heat output into the hot reservoir to the work input per cycle, we define coefficient of performance (for heating):
$$K_{h} = \frac{Q_{h}}{W_{\text{in}}} = \frac{Q_{h}}{Q_{h} – Q_{c}}$$
In the cooling mode, where we are interested in the ratio of heat extracted from the cold reservoir to the work input per cycle, we define coefficient of performance (for cooling):
$$K_{c} = \frac{Q_{c}}{W_{\text{in}}} = \frac{Q_{c}}{Q_{h} – Q_{c}}$$