Solids are made of atoms which are held together by intramolecular bonds to form molecules. The lattice of molecules are held together by intermolecular bonds and are arranged in cerain structures to form the solid.
There are 4 types of bonds holding lattices of molecules together:
- Covalent (chemical bond)
- Ionic (chemical bond)
- Metallic (chemical bond)
- Weak bonds (hydrogen, Van Der Waals etc)
Weak bonds are not permanent. They are merely brief attachments which can be broken down readily by a molecular collision. Strong bonds (covalent, ionic and metallic bonds) are almost never broken simply by molecular collision. They require a chemical action to break down.
Covalent Bonds
- Occurs between atoms and between molecules
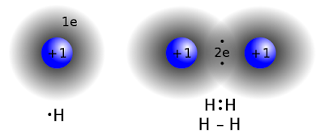
- Involves the sharing of electrons, usually with those shared electrons localized between two atoms.
- A bond can only be formed when the spins of the electrons are opposite. If the two electrons have parallel spins, they would have the same quantum numbers, which violates the Pauli Exclusion Principle -“No two electrons can ever occupy the same quantum state”.
- With opposite spins, the two electrons are in different quantum states and hence they can come together spatially. The region which the electrons spend much of their time in is between the two positively charged nuclei. Hence, the nuclei are attracted to the negativiy charged electron cloud. This attraction holds the atoms together forming a covalent bond in the molecule.
According to Heisenberg’s Uncertainty Principle:
- With a covalent bond, the electrons of the two atoms have a larger region of space to move about.(The electron cloud of both atoms merge)
- Hence, the uncertainty of the position of the electron (Δx) increases.
- Thus, the Δp decreases which leads to a decrease in E.
- Hence, it is more stable.(bond favourable)
Ionic Bonds
An ionic bond also occurs between atoms atoms and between molecules. It is a special case of a covalent bond. Instead of electrons being shared equally, they are shared unequally. Ionic bonds occur when an atom donates an electron to another atom. The donor becomes a positive ion (or cation), while the acceptor becomes a negative ion (or anion). Ionic bonds arise from the Coulomb (or electrostatic) force between positive cations and negative anions. (Example: Na+ and Cl– in a NaCl molecule)
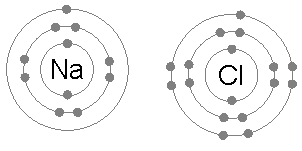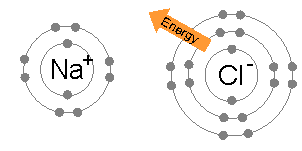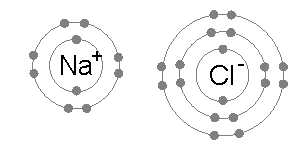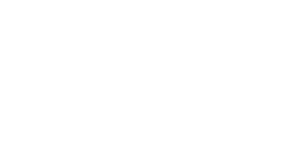
The electronic structure of Na is 1s22s22p63s1. A neutral sodium atom contains 11 electrons, 10 of which form closed shells (1s22s22p6) and shield the nucleus. Hence, the last electron in the 3s shell, which lies beyond these closed shells, experiences attraction by a net charge of +1e.
On the other hand, the electronic structure of Cl is 1s22s22p63s23p5. A neutral chlorine atom contains 17 electrons, 12 of which form closed shells (1s22s22p63s2) and shield the nucleus. Hence, Na would share its 3s valence electron with chlorine’s single 3p valence electron, implying covalent bonding. However, because its nucleus contains more protons, chlorine has a much greater affinity for electrons than sodium. It is more electronegative. As a result, the chlorine atom takes the lion’s share of the electron pair. The electron is essentially transferred from the Na atom to the Cl atom. We end up with a Na+ ion joined to a Cl– ion forming Na+Cl–.
Metallic Bonds
Metal atoms typically contain a high number of electrons in their outermost shell. The electrons become delocalised and form a sea of delocalised electrons surrounding a giant lattice of positive ions. The metal is held together by the strong forces of attraction between the positive nuclei and the delocalised electrons.
Metallic Bonding:
Metallic Bonding is the bonding within metals. it involves the delocalized sharing of free electrons among a lattice of metal atoms. Metal atoms typically contain a high number of electrons in their outermost shell. These become delocalized and form a sea of electrons surrounding a giant lattice of positive ions.
Sodium has the electronic structure 1s22s22p63s1. When sodium atoms come together, the electron in the 3s atomic orbital of one sodium atom shares space with the corresponding electron on a neighbouring atom, similar to the way that a covalent bond is formed.
The difference, however, is that each sodium atom is connected to eight other sodium atoms – and the sharing occurs between the central atom and the 3s orbitals on all of the eight other atoms. And each of these eight is in turn connected to another eight sodium atoms and so on, until all the atoms form a lump of sodium. All of the 3s orbitals on all of the atoms overlap to give a vast number of molecular orbitals which extend over the whole piece of metal.
The electrons can move freely within these molecular orbitals, and so each electron become delocalized from its parent atom. The metal is held together by the strong forces of attraction between the positive nuclei and the delocalised electrons.
Van Der Waal’s Bonds & Hydrogen Bonds
Summary:
- Van der waals bonds refer to the attachment between molecules with permanent dipole moments or induced dipole moments due to simple electrostatic attraction.
- When one of the atoms involved in a weak bond is hydrogen, it is called a hydrogen bond.
Explanation:
The binding energy for covalent and ionic bonds is typically 2 to 5eV. As such, these bonds are often known as strong bonds. Weak bonds, otherwise known as Van der Waals bonds, refer to attachment between molecules due to simple electrostatic attraction. van der Waals bonds are usually the result of attraction between dipoles. This can be between molecules with permanent dipole moments, or induced dipole moments.
When one of the atoms involved in a weak bond is hydrogen, it is called a hydrogen bond. A hydrogen bond is considered ‘strong’ for a weak bond. This is because the hydrogen atom is the smallest atom and thus can be approached more closely. A hydrogen bond also has partial ‘covalent’ character, thus making it stronger.
Hydrogen bonding is critical for life on Earth. For example, it is responsible for zipping together the double helix of DNA. Also, if water molecules were linked by Van der Waal’s forces alone, water would boil at about 200K. The hydrogen bonds between neighbouring water molecules are what raises the boiling point and allows water to be liquid at temperatures that prevail on Earth.