Table of Contents
The detection of radioactive emissions is a crucial process in various fields, including medical diagnostics, environmental monitoring, and nuclear physics. To accurately detect and measure these emissions, several types of detectors have been developed, each with its unique mechanism of action. Below, we expand and provide more insight into the common detectors used for this purpose.
Photographic detectors
Photographic detectors leverage the principle that radioactive emissions can affect photographic films in a manner akin to visible light. These detectors consist of films coated with silver halide, a compound sensitive to ionizing radiation. When exposed to radioactive substances, the silver halide crystals are altered, creating a latent image that captures the intensity and distribution of the radiation exposure. This latent image is then revealed through a chemical development process, allowing for the visualization and quantification of the radioactive emissions. This method is particularly useful for mapping radiation patterns and for applications requiring a permanent record of radiation exposure.
Geiger-Muller Tube (GM-tube)
Main article here: https://www.miniphysics.com/geiger-muller-tubecounter.html
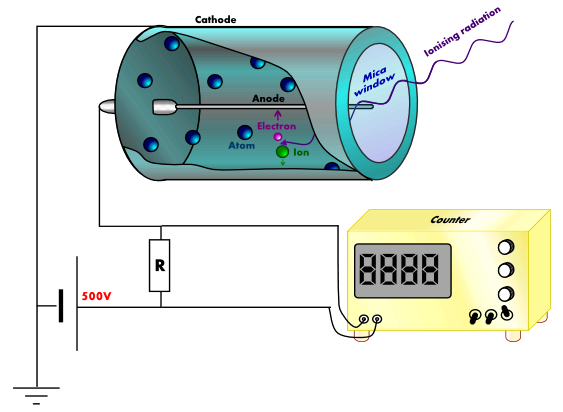
A GM-tube is the most useful radiation detector used for determining the intensity of a beam of radiation or for counting individual charge particles.
When ionizing radiation enters the tube through the thin mica window, it collides with the large argon atoms and ionized them.
These free electrons will then accelerate towards the fine wire anode placed along the axis of the cylindrical cathode. These accelerated electrons will cause further ionization of the argon atoms by colliding with them, thus producing an ‘avalanche’ of electrons which are collected almost at once by the anode.
The positively charged ions will be attracted towards the cathode. The collection of the electrons and the argon ions at the electrodes produces a current pulse. This current pulse is then amplified and fed to a ratemeter.
GM tubes are versatile, capable of detecting a wide range of radiation types, including alpha, beta, and gamma rays, making them indispensable in radiation monitoring and safety applications.
Diffusion Cloud Chamber

When emissions from the radioactive substance are allowed to come into contact with air molecules, it will tend to knock out the electrons of the molecules along its paths, causing ionization (i.e., the air molecules are electrically imbalanced and become ions).
The diffusion cloud chamber is the device for making visible the paths of ionizing emissions.
It consists of a container containing air. At the top of the chamber, a row of felt strips is soaked in ethanol (alcohol). The lower part of the chamber is cooled by solid carbon dioxide or liquid helium. The alcohol evaporates into vapour and continuously diffuses downwards. It then becomes supersaturated at the centre of the chamber (i.e., the air holds more vapour than it normally could).
When emissions from the radioactive substances are placed in the chamber, the emissions will cause ionization along its paths. The ionized air molecules will attract the vapour molecules and become too heavy to stay in suspension and thus, condense into tiny liquid droplets along the paths of the emissions.
This method allows for the direct observation of the paths of different types of radiation, with alpha particles creating thick, short tracks, and beta particles producing long, thin ones. The diffusion cloud chamber not only serves as an educational tool but also as a research instrument in particle physics.
Video Of A Diffusion Cloud Chamber:
Summary Table
| Detector Type | Description | Advantages | Disadvantages |
|---|---|---|---|
| Photographic Detectors | Uses films coated with silver halide sensitive to ionizing radiation, creating a latent image after exposure. |
|
|
| Geiger-Müller Tube | Measures radiation by ionization of gas within a tube, causing an electrical pulse. |
|
|
| Diffusion Cloud Chamber | Visualizes the paths of ionizing radiation through the formation of droplets along their path in a supersaturated environment. |
|
|
Worked Examples
Example 1: Understanding Photographic Detectors
A laboratory technician uses a photographic film detector to measure the distribution of a radioactive source. After developing the film, they notice that some areas are much darker than others. What does this indicate about the radioactive source, and how might the technician use this information?
Click here to show/hide answer
The darker areas on the photographic film indicate regions of higher radiation intensity, as more radiation has interacted with the silver halide crystals in those areas, leading to a greater degree of exposure. The technician can use this information to map the radiation field of the source, identifying areas of higher and lower activity. This could be crucial for applications such as radiation therapy planning, where ensuring the accurate delivery of radiation doses is vital.
Example 2: Geiger-Müller Tube Practicality
A field researcher uses a Geiger-Müller (GM) tube to monitor environmental radiation levels in various locations. At one site, the GM tube registers 120 counts per minute (cpm), while at another, it records 20 cpm. What might these readings suggest about the two locations, and what precautions should the researcher take?
Click here to show/hide answer
The site with 120 cpm exhibits a significantly higher level of radiation compared to the site with 20 cpm. This suggests that the first site may have higher radioactive material presence or contamination. The researcher should take appropriate safety precautions, such as minimizing exposure time, maintaining a safe distance, and using protective gear when in the higher radiation area. Further investigation might be needed to determine the source of radiation and assess any environmental or health risks.
Example 3: Diffusion Cloud Chamber Observations
During a demonstration with a diffusion cloud chamber, a student observes short, thick tracks and long, thin tracks. What types of particles are likely causing these tracks, and what does their appearance tell us about the properties of these particles?
Click here to show/hide answer
The short, thick tracks are likely caused by alpha particles, which are heavier and carry more charge than beta particles, resulting in more substantial ionization along their path. The long, thin tracks are indicative of beta particles, which are lighter and less charged, leading to less ionization and therefore a thinner track. This observation tells us that alpha particles have a greater ionizing power but a shorter range in air compared to beta particles, which have a longer range but lower ionizing power.
Example 4: Comparing Detector Efficiencies
A scientist has access to a photographic film detector, a GM tube, and a diffusion cloud chamber. If they aim to conduct a comprehensive study on the nature and intensity of radiation from an unknown source, how should they utilize each detector to maximize their understanding?
Click here to show/hide answer
To maximize understanding, the scientist should use each detector to leverage its strengths:
- Photographic Film Detector: Use this to get a permanent record of the radiation’s spatial distribution. It’s especially useful for observing the overall exposure pattern and identifying areas with higher radiation intensity.
- GM Tube: Utilize the GM tube for precise measurements of radiation intensity across different points. It’s ideal for quantifying the radiation levels and detecting variations in radiation intensity with high sensitivity.
- Diffusion Cloud Chamber: Employ the cloud chamber to visually observe the types of radiation (alpha, beta, etc.) and understand their properties, such as range and ionization capability. It’s excellent for qualitative analysis and identifying the nature of the radioactive emissions.
By combining these approaches, the scientist can obtain a detailed profile of the radiation, including its distribution, intensity, and the types of particles emitted.
Example 5: Safety and Radiation Detection
A safety officer at a nuclear facility needs to ensure workers are not exposed to harmful levels of radiation. They decide to use a GM tube for routine checks and a photographic film badge for long-term monitoring. Explain how each method contributes to the safety of the workers and the advantages of using both.
Click here to show/hide answer
- GM Tube for Routine Checks: The GM tube provides immediate feedback on radiation levels, allowing the safety officer to quickly identify and respond to areas with unsafe radiation levels. It’s ideal for day-to-day monitoring and ensuring that workers are not currently exposed to harmful radiation as they move around the facility.
- Photographic Film Badge for Long-term Monitoring: The film badge records the cumulative exposure of each worker over time. This is crucial for assessing long-term radiation exposure and ensuring it does not exceed safe limits. The badge can show variations in exposure, identifying workers or areas that may require further safety measures.
Using both methods offers a comprehensive safety approach, combining real-time detection with cumulative exposure monitoring. This ensures both immediate and long-term radiation safety for the workers, addressing both acute and chronic exposure risks.