Table of Contents
Boiling
Boiling is the change of state from a liquid to a gas. Boiling of a pure substance occurs at a particular constant temperature called boiling point or boiling temperature.
- During boiling, the molecules need energy to overcome the intermolecular forces that hold them together and to increase the separation between the molecules. Some energy is also required to allow the vapour to expand against atmospheric pressure. The total energy required is the latent heat of vaporisation.
Condensation
The change of state from a gas to a liquid is called condensation. A pure substance condenses at a temperature equal to its boiling point.
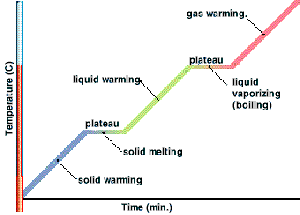
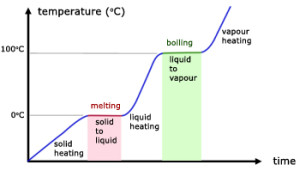
From the graph above,
- $100^{\circ}\text{C}$ is the boiling point of the liquid
- Temperature remains constant at $100^{\circ}\text{C}$ as the liquid is boiling
- During the boiling process, liquid and gas exist in equilibrium.
Step-By-Step Of What Happens During Boiling
- Heat energy is absorbed by the particles
- The kinetic energy of the particles increases
- At boiling point, the particles have enough energy to overcome the attractive forces between the particles and move away from one another.
- The particles escape from the liquid to form gas
Cause For Constant Temperature During Boiling
The absorbed heat energy is used to overcome the attractive forces between the particles and not the kinetic energy.
Factors That Affect Boiling Point
Adding impurities to a liquid affect its boiling point. E.g. By adding a 100 g of salt to 1000 g of water increases its boiling point by $1^{\circ}\text{C}$
When the liquid vapourises, volume of the liquid expands. High pressure applied to liquid will oppose its expansion into gas and thus, opposes boiling. Therefore, the boiling point of the liquids increases with increasing pressure. A reduction in pressure lowers the boiling point.
- In mountains, water boils at lower temperatures as the atmospheric pressure decreases with the elevation.
- Evaporated milk is produced by boiling the milk under reduced pressure. Boiling under reduced pressure saves energy required to boil and the lowered temperature preserves the vitamins in the milk better.
- In pressure cookers, when the cooking pressure increases and the boiling point of water inside the cooker increases. Therefore, higher temperature can be achieved and the food can be cooked more quickly.
Note: The boiling point of pure water is 100 °C at standard atmospheric pressure.
Evaporation
The process in which liquid change into vapour without boiling is called evaporation. It occurs at all temperatures.
- Evaporation is the change of state, from liquid to gas, that takes place at the surface of a liquid at any temperature.
At home, wet clothes are dried by evaporation. The water in the clothes is unlikely to reach its boiling point even though the clothes are hung under the hot sun. The drying of clothes is only by the evaporation process.
Process Of Evaporation
In liquid, the speeds of molecules in random motion are different and keep changing due to the intermolecular bombardments. When a molecule near the surface of the liquid gets sufficient speed or energy, it escapes from the liquid surface and leaves behind a liquid with a lower energy content (= lower temperature). This is effectively cooling by evaporation.
Factors Affecting Speed Of Evaporation
Evaporation could occur at any temperatures. However, the following INCREASES the speed of evaporation:
- Higher temperature: At higher temperatures, the evaporation process speeds up as there will be more particles in the liquid that are moving fast enough to escape from the surface
- Larger surface area: With a larger liquid surface area, there will be more particles which have a change to escape from the liquid surface.
- Having a wind blowing above the liquid surface: A wind blowing over the surface will carry the vapour particles away from the liquid surface, which stops them from returning to the liquid and making it easier for more liquid particles to escape from the liquid surface.
- Lower air/atmosphere humidity: A lower humidity will have the same explanation as above whereby there will be less vapour particles above the liquid surface which makes it easier for more liquid particles to escape from the liquid surface.
- Lower air/atmosphere pressure: A lower pressure will have the same explanation as above whereby there will be less vapour particles above the liquid surface which makes it easier for more liquid particles to escape from the liquid surface.
- Lower boiling point of liquid: If the boiling point of the liquid is lower, the particles need LESS energy to escape from the liquid surface and hence it is easier for more liquid particles to escape from the liquid surface.
Rate if evaporation increases with temperature, area of the exposed surface and wind; and, decreases with humidity of the atmosphere, pressure and boiling point of the liquid.
Uses Of Cooling By Evaporation
Sweating results in the evaporation of water from the skin, serving as the body’s mechanism for dissipating excess heat and maintaining a consistent temperature. Engaging in intense physical activity raises the potential for the body to become excessively cooled, particularly in drafty conditions, thereby reducing its ability to fend off infections.
Ether functions as a local anesthetic by cooling (as well as cleansing) the arm during an injection.
Large-scale cooling through evaporation is employed in refrigerators, freezers, and air-conditioning systems.
Perfumes utilize volatile liquids for their fragrance.
Difference Between Boiling & Evaporation
The table below illustrates the differences between boiling and evaporation.
| Boiling | Evaporation |
|---|---|
| A process in which a substance changes its state from the liquid state to the gaseous state | A process in which a substance changes its state from the liquid state to the gaseous state without boiling |
| Fast | Slow |
| Bubbles are formed | No bubbles formed |
| Occurs throughout the liquid | Takes place only from the exposed surface of the liquid |
| Occurs at a definite temperature – Boiling point | Occurs at all temperature |
| Source of energy needed | Energy supplied by surroundings |
Worked Examples
Example 1
a. When a solid undergoes melting:
i. Does its temperature rise, fall, or stay constant?
ii. Is energy absorbed, released, or neither?
iii. Does the kinetic energy of the particles rise, fall, or stay constant?
b. When a liquid is undergoing boiling:
i. Does its temperature rise, fall, or remain constant?
ii. Does the potential energy of the particles increase, decrease, or remain constant?
Click here to show/hide answer
a. When a solid undergoes melting:
i. The temperature remains constant because the energy supplied is utilized to overcome the forces holding the particles together, allowing them to transition from a solid to a liquid state.
ii. Energy is absorbed during melting as the solid absorbs heat to break the bonds between its particles and facilitate the phase transition.
iii. The kinetic energy of the particles increases as they gain the energy needed to move more freely in the liquid state.
b. When a liquid is undergoing boiling:
i. The temperature remains constant during boiling as the supplied heat is used to convert the liquid into vapor rather than increase the temperature.
ii. The potential energy of the particles increases during boiling, as they absorb heat energy to overcome intermolecular forces and transition from the liquid to the gaseous state.
Example 2
a. Explain the particle-level details of the evaporation process.
b. What happens to the temperature of a liquid as it undergoes evaporation?
Click here to show/hide answer
a. The process of evaporation at the particle level involves individual liquid particles gaining enough energy to break their intermolecular bonds and transform into a gaseous state. As these particles absorb heat energy, their kinetic energy increases, allowing them to overcome attractive forces within the liquid. Eventually, some particles near the liquid’s surface gain sufficient energy to escape into the air as vapor. This continuous process leads to the gradual conversion of liquid to vapor.
b. During evaporation, the temperature of the liquid remains constant. The absorbed heat energy is utilized to break the intermolecular forces and facilitate the phase transition from liquid to gas, rather than causing an increase in temperature. This phenomenon is due to the fact that the absorbed energy contributes to the potential energy of the particles, enabling them to escape the liquid phase without a corresponding rise in temperature.
Example 3
A portion of water is contained in a permeable bag, like canvas, suspended in an area open to a stream of air. Clarify why the water’s temperature is cooler compared to the surrounding air.
Click here to show/hide answer
The water inside the permeable bag, such as canvas, is cooler than the surrounding air due to the process of evaporative cooling. When the bag is exposed to a draft of air, the water molecules at the surface of the bag gain energy from the air and undergo the process of evaporation. As these water molecules transition from liquid to vapor, they absorb heat energy from the surroundings, including the water inside the bag. This absorption of heat energy leads to a reduction in the temperature of the water, making it cooler than the surrounding air. Essentially, the evaporation of water acts as a cooling mechanism, and in this context, it causes the water stored in the porous bag to be at a lower temperature than the air around it.
Example 4
How come engineers frequently utilize superheated steam (steam at temperatures exceeding 100°C) for heat transfer?
Click here to show/hide answer
Firstly, superheated steam carries more energy compared to normal steam. Superheated steam, being at a temperature (exceeding 100°C) above the boiling point, has absorbed more heat energy compared to normal steam at the same pressure. This additional energy is stored as sensible heat in the form of higher temperature. As a result, superheated steam carries more energy per unit mass than normal steam at the same pressure.
With the higher temperature, there will be a greater temperature differential between the superheated steam and the substance being heated. A higher temperature differential enhances heat transfer rates, making the process more efficient.
As superheated steam is at a much higher temperature than its boiling point, it can lose more energy before condensing. Condensation is a concern because it may lead to potential issues like erosion in pipes.