Table of Contents
Atom: The Core Of Matter
Matter is fundamentally composed of tiny particles known as molecules, which are in perpetual motion. These molecules are made up of smaller units called atoms, which are the basic building blocks of all chemical elements.
- The atom is considered the smallest unit of matter that retains the properties of an element, making it the foundation of chemistry.
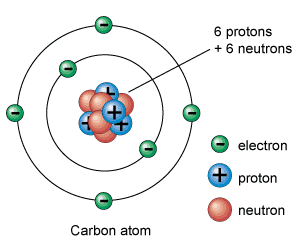
An atom is distinguished by its composition of a small, dense nucleus, surrounded by a cloud of electrons. This nucleus, containing protons and neutrons, is positively charged and makes up nearly all the atom’s mass despite occupying a minuscule volume of its space.
Nucleus: The Dense Heart
Each atom consists of a nucleus and electrons. A nucleus is a compact core of an atom and it consists of particles called protons and neutrons.
Atoms are mostly empty space. The nucleus contains 99.95% of the atom’s mass but only a tenth-billionth part of its volume. Practically all the mass of an atom is the mass of its nucleus.
Protons & Neutrons
Protons & neutrons are known as nucleons as they are found in the nucleus. Protons & neutrons have almost the same mass.
A proton, a constituent of every atomic nucleus, is a stable sub-atomic particle that has a unit-positive electric charge (i.e. 1.60 x 10-19 C) and a rest mass of 1.67 x 10-27 kg.
A neutron, a constituent of every atomic nucleus except ordinary hydrogen, is a stable sub-atomic particle that has no electric charge and a rest mass of 1.67 x 10-27 kg.
Do you know?
The proton and the neutrons are made of much smaller particles, called quarks. You might ask: are they themselves made by even smaller particles? Physicists are proposing that everything in the Universe might be made up of tiny, vibrating closed strings. If you are interested, do a google search on string theory.
Electrons: The Dynamic Cloud
The space surrounding the nucleus is occupied by the electrons. The electrons orbit the neucleus and are moving so rapidly that we can think of them as being everywhere at once. (Note: This is not very accurate, but it will suffice for O Level.)
An electron is not part of the atomic nucleus, but orbits the nucleus. It carries a unit-negative electric charge (i.e. -1.60 x 10-19 C). An electron is nearly mass-less. It has a rest mass of 9.11 x 10-31 kg. The rest mass of a proton is 1836 times the rest mass of an electron.
In a neutral atom, the number of electrons is the same as the number of protons. Circulating electrons are held in orbit by the influence of protons’ positive charge.
Do you know?
The electron has an anti-matter counterpart called the positron. This anti-particle has precisely the same mass, but it carries a positive charge. If it meets an electron, both are annihilated in a burst of energy.
The hydrogen nucleus is unique in having no neutron, only a single proton.
Proton Number & Nucleon Number
All naturally-occurring materials are made up from chemical elements. (hydrogen, helium, etc)
Atoms of different chemical element have a different numbers of protons in their nuclei, and therefore different numbers of electrons in the orbit. Chemical elements are characterized and identified by their atomic numbers and have unique chemical properties.
The proton (or atomic) number is the number of protons present in the nucleus of the atom.
- The atom is electrically neutral. Hence, the atom has the same number of protons as its number of electrons.
The nucleon (or mass) number is the number of nucleons (sum of protons and neutrons) present in the nucleus of the atom.
- Since the mass of an atom is concentrated at its nucleus, the nucleon (or mass) number is thus an approximate measure of the mass of the atom.
- The number of neutrons present in the nucleus of the atom (the neutron number, N) may be deduced by subtracting the proton number (Z) from its nucleon number (A).
Nucleon number (A) = neutron number (N) + proton number (Z)
Isotopes: Variations Of Element
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons, leading to variations in mass.
- Mass of the isotopes of an element differs as the number of neutrons varies.
- The number or protons in the nucleus of an atom determines what chemical element it is.
- Every chemical element has one or more isotopes. An atom is first identified and labeled according to the number of protons in the nucleus.
These differences do not affect the chemical properties of the atoms, as chemical reactions involve only the electrons. However, isotopes exhibit distinct nuclear and physical properties
- Chemical reactions only involve electrons. The protons and neutrons in the nucleus take no part in chemical reactions.
- All atoms with the same electron arrangement, hence, same proton number, have nearly, if not precisely, identical chemical properties, but with different nuclear and physical properties.
Understanding Chemical Element Symbols: A Guide To Isotopic Notation
Writing the symbol for a chemical element in its isotopic form involves a notation that provides specific information about the element’s atomic number, mass number, and sometimes its charge. This notation is crucial in various scientific fields, including chemistry, physics, and biology, as it conveys detailed information about the atom’s composition in a compact form. Here’s a breakdown of how to write a chemical element’s symbol, using helium as an example:
Basic Format: $\text{X}^A_Z$
- $X$: The chemical symbol of the element (e.g., $\text{He}$ for helium). This symbol is a one or two-letter abbreviation of the element’s name in English (or Latin in some cases), with the first letter capitalized and the second letter in lowercase.
- $A$: The mass number (also known as the nucleon number) of the atom, which is the total number of protons and neutrons in the nucleus. For helium-4, this number is $4$. The mass number is written as a superscript to the upper left of the chemical symbol.
- $Z$: The atomic number of the element, which is the number of protons in the nucleus. This number defines the element’s identity in the periodic table. For helium, the atomic number is $2$. The atomic number is written as a subscript to the lower left of the chemical symbol.
Example: Helium-4
For helium-4, the symbol is written as $\text{He}^{4}_2$. This notation indicates that the helium atom has:
- 2 protons (as indicated by the atomic number, $Z = 2$)
- A total atomic mass of 4 (indicating that there are 2 neutrons in addition to the 2 protons, since $4-2 = 2$ neutrons)
Isotopes & Ions
- Isotopes: Different isotopes of an element have the same atomic number ($Z$) but different mass numbers ($A$). For example, hydrogen has three common isotopes: $\text{H}^{1}_1$ (protium), $\text{H}^{2}_1$ (deuterium), and $\text{H}^{3}_1$ (tritium).
- Ions (more applicable to Chemistry): If the atom is an ion (i.e., it has a net electric charge due to the loss or gain of electrons), the charge can be written as a superscript to the upper right of the element symbol. For example, a positively charged helium ion with two protons and no electrons (a helium nucleus) would be written as $\text{He}^{2+}$, but often the mass number and atomic number are omitted for ions unless specifically discussing isotopes.
This notation system allows scientists to convey detailed information about the atomic structure of elements and their isotopes quickly and efficiently, providing insight into their physical and chemical properties.
Worked Examples To Test Your Understanding
Example 1: Understanding Atomic Structure
Consider an atom of carbon with 6 protons in its nucleus. If this atom is electrically neutral, how many electrons does it have, and what is its nucleon number if it contains 6 neutrons?
Click here to show/hide answer
An electrically neutral atom has an equal number of protons and electrons. Therefore, a carbon atom with 6 protons also has 6 electrons. The nucleon number, or mass number, is the sum of protons and neutrons. With 6 protons and 6 neutrons, the nucleon number is $6 + 6 = 12$.
Example 2: Protons, Neutrons, and Electrons
If an oxygen atom has 8 electrons and 8 protons, what is its atomic number, and how many neutrons does it have if its nucleon number is 16?
Click here to show/hide answer
The atomic number is equal to the number of protons, so for oxygen with 8 protons, the atomic number is 8. The nucleon number is 16, which is the sum of protons and neutrons. Therefore, the number of neutrons is $16 – 8 = 8$.
Example 3: Identifying Isotopes
Hydrogen has three isotopes: protium with no neutron, deuterium with one neutron, and tritium with two neutrons. If all three isotopes have one proton, what are their nucleon numbers?
Click here to show/hide answer
- Protium has 1 proton and no neutrons, so its nucleon number is $1 + 0 = 1$.
- Deuterium has 1 proton and 1 neutron, so its nucleon number is $1 + 1 = 2$.
- Tritium has 1 proton and 2 neutrons, so its nucleon number is $1 + 2 = 3$.
Example 4: Calculating Electron Arrangement
An ion of nitrogen has a charge of -3. Given that a neutral nitrogen atom has 7 protons, how many electrons does this ion have?
Click here to show/hide answer
A neutral nitrogen atom has 7 electrons, equal to its number of protons. If the nitrogen ion has a charge of -3, it means it has 3 extra electrons, making the total number of electrons (7 + 3 = 10).
Example 5: Distinguishing Isotopes by Mass Number
Carbon-12 and Carbon-14 are two isotopes of carbon differing in their number of neutrons. Given that both isotopes have 6 protons, how many neutrons does each isotope have, and how does this difference impact their chemical & physical properties?
Click here to show/hide answer
- Carbon-12 has 6 neutrons because its nucleon number is 12 and it has 6 protons ($12 – 6 = 6$).
- Carbon-14 has 8 neutrons because its nucleon number is 14 and it has 6 protons ($14 – 6 = 8$).
The chemical properties of isotopes of an element are generally the same because chemical properties are primarily determined by the number of electrons and the arrangement of these electrons around the nucleus. Since both carbon-12 and carbon-14 have 6 protons, they will also have 6 electrons in a neutral state, leading to identical chemical behavior. This is why both isotopes of carbon participate in the same chemical reactions in the same way.
The difference in the number of neutrons, however, does have a significant impact on the physical properties of the isotopes:
- Mass: Carbon-14 is heavier than carbon-12 due to the two additional neutrons. This difference in mass can affect the isotope’s behavior in physical processes, such as diffusion or sedimentation.
- Radioactivity: Carbon-14 is radioactive, meaning it undergoes decay over time, transforming into nitrogen-14 through beta decay. This property makes carbon-14 useful in radiocarbon dating of archaeological and geological samples. Carbon-12, on the other hand, is stable and does not undergo radioactive decay.
- Stability and Abundance: Carbon-12 is the more abundant isotope of carbon found in nature, making up about 98.9% of all carbon. Its stability contributes to its abundance, as it does not decay over time. Carbon-14, while naturally occurring, is much less abundant and is continuously produced in the upper atmosphere through the interaction of nitrogen atoms with cosmic rays.