The binding energy per nucleon of a nucleus is the binding energy divided by the total number of nucleons.
- Measure of stability of the nucleus. Larger the binding energy per nucleon, the greater the work that must be done to remove the nucleon from the nucleus, the more stable the nucleus
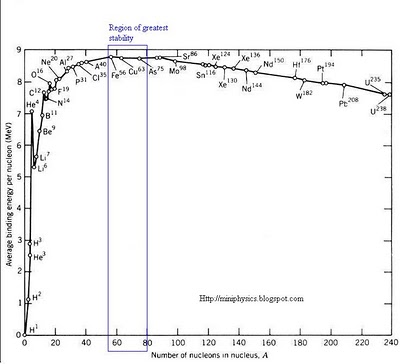
Graph of the variation of binding energy per nucleon with nucleon number
Important features of the graph:
- Excluding the lighter nuclei, the average binding energy per nucleon is about 8 MeV.
- The maximum binding energy per nucleon occurs at around mass number A = 50, and corresponds to the most stable nuclei. Iron nucleus Fe56 is located close to the peak with a binding energy per nucleon value of approximately 8.8 MeV. It’s one of the most stable nuclides that exist.
- Nuclei with very low or very high mass numbers have lesser binding energy per nucleon and are less stable because the lesser the binding energy per nucleon, the easier it is to separate the nucleus into its constituent nucleons.
- Nuclei with low mass numbers may undergo nuclear fusion, where light nuclei are joined together under certain conditions so that the final product may have a greater binding energy per nucleon.
- Nuclei with high mass numbers may undergo nuclear fission, where the nucleus split to give two daughter nuclei with the release of neutrons. The daughter nuclei will possess a greater binding energy per nucleon.