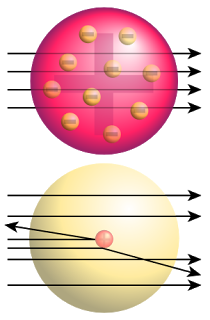
J.J. Thomson proposed a model of the atom – Plum-pudding model of the atom
Characteristic of plum-pudding model of the atom
- Positive charge of the atom is spread out through the entire volume of the atom
- Electrons are distributed throughout this volume
- Electrons vibrate about their equilibrium positions within the sphere of charge
The ɑ-particle scattering experiment
Using a beam of positively charged alpha particles to fire on a thin gold foil
Video of the experiment:
- ɑ-particles carry a charge of +2e and are about 7300 more massive than electrons.
- If Thomson’s model was correct, maximum deflecting force on the ɑ-particle as it passes near a positive charge will be far too small to deflect the particle by even 1o.
- Electrons in the atom would also have very little effect on the massive, energetic ɑ-particle.
Actual Results:
- Most of the particles went straight through or were scattered through very small angles.
- But a very small fraction (less than 1%) of the particles were scattered through very large angles, some of which were close to 180o.
Interpretation of the actual results:
1. Large deflection of ɑ-particles
- To produce such a large deflection, there must be a large force.
- Such a large force is only possible if we assume that the atom consists of a positively charged nucleus of very small dimensions compared with the ordinarily accepted magnitude of the diameter of the atom.
- When an ɑ-particles gets close to the centre of the concentration of positive charges(nucleus), it cannot penetrate the nucleus but gets deflected instead due to the large repulsive force between it and the positively charged nucleus.
2. Most particles passed through undeflected or with a small deflection
- Since very few of the particles were scattered through large angles, the probability of the particle getting close to the centre of the positive charge is small. This shows that the nucleus occupies only a small proportion of the available space.
3. Few particles are deflected backwards, through an angle close to 180o.
- The nucleus is small and very massive.
Simulation of the experiment: http://phet.colorado.edu/en/simulation/rutherford-scattering