Bohr Model of the atom
Assumptions:
- An atom consists of negative electrons orbiting a very small positive nucleus. The electron moves in circular orbits around the nucleus under the influence of the electric force of attraction between the electron and the positively charged nucleus.
- The electrons can only take certain orbits as only certain orbits are stable. When an electron is in such an orbit, it is said to be in a stationary state, and thus will not emit energy in the form of radiation.(In Rutherford’s model, electron experience acceleration, hence emitting electromagnetic radiation, losing energy and spiralling down to the nucleus)
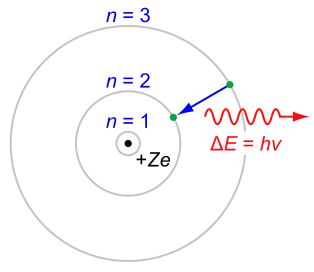
- Atoms can only exist in stationary states(energy level/shell). Each stationary state corresponds to a particular set of electron orbits around the nucleus. These states are distinct and can be numbered as n = 1,2,3,4,… where n is the quantum number.
- Each stationary state has a discrete, well-defined energy En. Atomic energies are quantized. The stationary states of an atom are numbered in order of increasing energy: E1 < E2 < E3 < E4
- The lowest energy state(most stable) of an atom with energy E1, is called the ground state of the atom. Other stationary states with energies E2, E3, E4, … are called excited states of the atom.
- An atom can “jump” from one stationary state to another by emitting or absorbing a photon with energy of photon equal to the energy difference between 2 levels before it can be absorbed. Otherwise, it will not be absorbed at all.
- An atom can also move from a lower energy state to a higher energy state by absorbing energy in an inelastic collision with an electron or another atom. This process is called collisional excitation. Colliding particle will impart energy equal to the difference in energy states and leave with any excess energy. Colliding particles can give the greatest amount of energy to help the orbital electron reach the highest possible energy state.
- Atoms will seek the lowest energy state. An atom in an excited state, if left along, will jump to lower and lower energy states unit it reaches the ground state.
Bohr model of the atom explained
- why matter is stable: Once an atom is in its ground state, there are no states of any lower energy to which it can jump. It can remain in the ground state forever.
- why atoms emit and absorb a discrete spectrum: Only those photons whose frequency match the energy intervals between the stationary states can be emitted or absorbed. Photons of other frequencies cannot be emitted or absorbed without violating energy conservation.
- why each element has a unique line spectrum: The energies of the stationary states are just the energies of the orbiting electrons. The atoms has no other forms of energy. Different elements, with different numbers of electrons, will have different stable orbits and thus different stationary states. States with different energies will emit and absorb photons of different wavelengths.