Assumption: Electromagnetic radiation is emitted in quanta and also absorbed in discrete units.
Since electrons are held by attractive forces to the atoms, to escape form a metal, an electron must do a certain amount of work to remove itself from the surface to infinity. An electron in a metal can only escape if it gains enough energy from a single photon to enable it to do the necessary work. So, individual photon must each supply more than a certain amount of energy.
The work function $\Phi$ of a material is defined as the minimum amount of the work necessary to remove a free electron from the surface of the material.
Einstein’s photoelectric equation: $E_{k \, max} = hf – \Phi$
- hf is the energy of the photon
- The electrons with maximum kinetic energy $E_{k \, max}$ come from the surface of the metal. Due to collisions with other atoms, those below the surface emerge with a ssmaller kinetic energy.
Threshold frequency is the minimum frequency of an incident radiation required to just remove an electron from the surface of a metal.
Threshold frequency( $f_{o}$) explained:
- Electrons can only escape if the maximum kinetic energy is greater than zero.
$\begin{aligned} E_{k \, max} &> 0 \\ hf – \Phi &> 0 \\ f_{o} &> \frac{\Phi}{h} \end{aligned}$
For photoelectric effect to take place, $f > f_{o}$.
Instantaneous emission of electrons explained:
- An electron is emitted if it gains enough energy from the photon. Since all photon energy is delivered immediately to the electron in a single collision, there is no time delay and is independent on the intensity of the incident radiation.
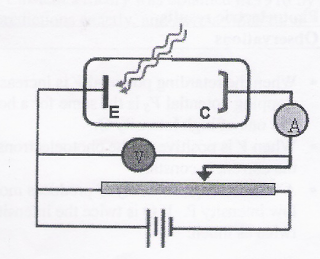
For a metal illuminated by electromagnetic wave,
- If the potential of E is held negative with respect to C, the photoelectrons escape more easily because they are repelled by the surface.
- But if potential of C is negative with respect to E, there exists a minimum negative potential required to stop even the most energetic electron from reaching electrode C.
The maximum kinetic energy $E_{k \, max}$ which a photoelectron can have:
$\begin{aligned} Loss \: in \: KE &= Gain \: in \: EPE \\ E_{k \, max} &= eV_{s} \\ &= \frac{1}{2} mv_{max}^{2} \\ &= eV_{s} \end{aligned}$
, where $V_{s}$ is the stopping potential
Substituting above equation into Einstein’s photoelectric equation,
$V_{s} = \frac{h}{e} f \, – \, \frac{\Phi}{e}$