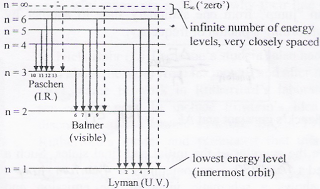
- n represents the principle quantum number and only takes integral values from 1 to infinity.
- The ground state refers to the lowest energy level n=1 in which the atom is the most stable. The electron normally occupies this level unless given sufficient energy to move up to a higher level. An atom is said to be in an excited state when its electrons are found in the higher energy levels. When an atom is excited from the ground state to a higher energy, it becomes unstable and falls back to one of the lower energy levels by emitting photon(s)/electromagnetic radiation
- The highest energy level n = ∞ corresponds to an energy state whereby the electron is no longer bound to the atom. (the electron has escaped from the atom.) By convention, it is usually assigned an energy value of 0 eV.
- The lower the energy level, the more negative the energy value associated with that level. Thus, the lower energy states correspond to more stable states.
- The energy difference between any two adjacent levels gets smaller as n increases, which results in the higher energy levels getting very close and crowded together just below n = ∞ .
- The ionization energy of an atom is the energy required to remove the electron completely from the atom.(transition from ground state n = 0 to infinity n = ∞). For hydrogen, the ionization energy = 13.6eV
- When an excited electron returns to a lower level, it loses an exact amount of energy by emitting a photon. The Lyman(ultraviolet) series of spectral lines corresponds to electron transitions from higher energy levels to level n = 1. Transitions to n = 2 and n = 3are called the Balmer(visible) and Paschen(Infra Red) series, respectively.
Why the energy levels have negative values?
- Negative value of energy indicates that the electron is bound to the nucleus and there exists an attractive force between the electron and the nucleus. Also, since the potential at infinity is defined as zero, energy levels at a distance below infinity are negative.