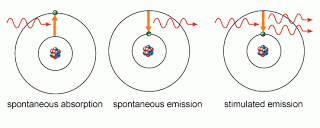
Atoms excite and de-excite by photons via 3 processes
- Stimulated absorption – An atom can be excited from a lower energy state(or ground state) E1 to a higher energy state E2 if one of it’s electron absorbs a photon of energy equal to the difference in energy E2 – E1.
- Spontaneous emission – An atom in an excited state can go to a lower energy state(or ground state) E1 by the spontaneous emission of a photon with energy equal to E2-E1
- Stimulated emission – An atom in an excited state can be stimulated by an incident photon to emit another photon of the same frequency, phase, momentum and in the same direction of travel as the incident photon.
Stimulated emission and spontaneous emission are competing with each other to take place. An excited electron can de-excite via stimulated emission or spontaneous emission.(depending on which process occurs faster)
- Spontaneous emission occurs more rapidly when the lifetime of the excited state is short and this is the case for electron undergoing high energy transitions. Henceforth, spontaneous emission competes more strongly with stimulated emission in transitions that emit blue(400nm) or ultraviolet light than for transitions that emit red (700nm) or infrared radiation.
- Stimulated emission occurs more rapidly when the stimulating photon beam is of high intensity.(a great number of incoming photons)
Electrons can be excited by bombardment by electrons. There is no need for the colliding electrons to have energy equal to the E2-E1, the colliding electrons will just leave with the excess energy.