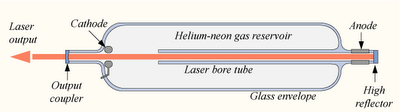
Helium-Neon Laser
- Emits wavelength of 632.8 nm.
- Consists of a mixture of ~90% Helium gas, ~10% Neon gas
- The gases are enclosed in a glass tube.
- An electrical discharge is established along the bore of the tube
- Two mirrors are bonded to the ends of the discharge tube, one a total reflector, the other having ~1 % transmission
Information you need for the next part:
A metastable state is an excited state of a system which has a much longer lifetime(~ 10-3s) than that of the normal excited states(~10-8).
Step by step laser action
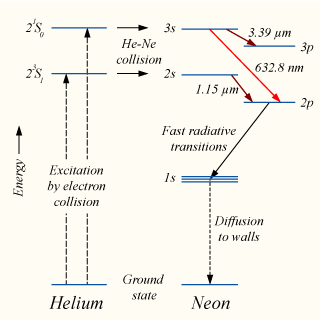
- A high voltage is applied across the tube to excite the helium atoms
- Collision of electrons from the electrical discharge and the helium atoms in the tube
- This excites the helium atoms from the ground state to the 23S1 and 23S0 metastable excited states.
- These excited He atoms cannot return to its ground state via the normal emission of photons. (due to violation of selection rules)
- The excited He atoms can only de-excite via collision with non-excited atoms.
- When the excited helium atoms collide with the ground-state neon atoms, there is a transfer of energy to the neon atoms, exciting them into the 2s and 3s states. The transfer(excitation transfer) is possible due to a coincidence of energy levels between the helium and neon atoms.
- The number of neon atoms entering the excited states builds up as further collisions between helium and neon atoms occur, causing a population inversion between the neon 3s and 2s, and 3p and 2p states.
- Spontaneous emission between the 3s and 2p states results in the emission of 632.8 nm wavelength light which in turn results in stimulated emission of 632.8 nm light.
- After this, fast radiative decay occurs from the 2p to the 1s energy levels.
- From the 1s energy levels, the neon atoms can only decay to the ground state via collision with container walls.
With other cavity mirrors, other wavelengths of laser emission of He-Ne lasers are possible too.
- Green – 543.5 nm
- Yellow – 594 nm
- Orange – 612 nm